Optimal design of clinical trials with computer simulation based on results of earlier trials, illustrated with a lipodystrophy trial in HIV patients - ScienceDirect
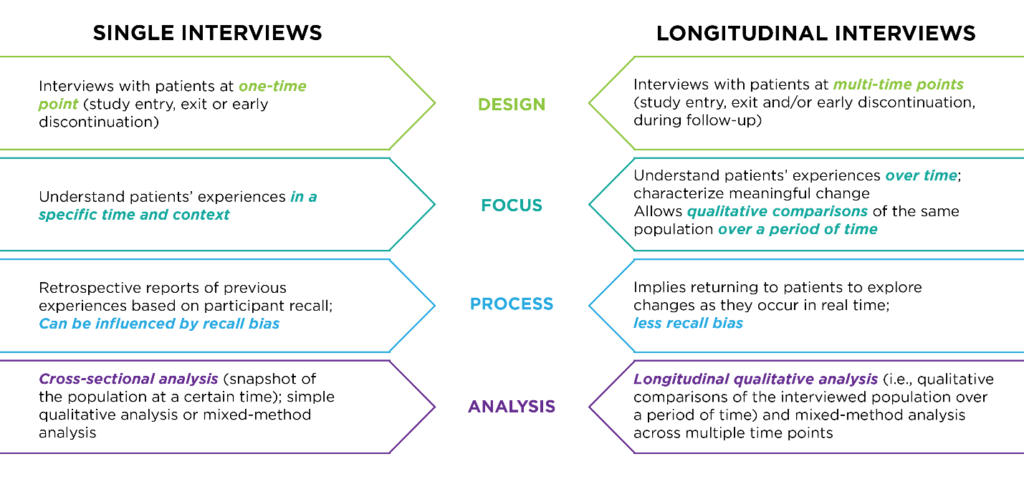
Guidance on Research Follow-Up Defining a follow-up period is important to the research protocol (even if the follow-up period i

Sustained Effects of Different Exercise Modalities on Physical and Mental Health in Patients With Coronary Artery Disease: A Randomized Clinical Trial - Canadian Journal of Cardiology

DAHLIA project overview including highlights of each study and time... | Download Scientific Diagram

Long-term follow-up of a prospective phase 2 clinical trial of extended treatment with rituximab in patients with B cell post-transplant lymphoproliferative disease and validation in real world patients | SpringerLink
Are cancer patients better off if they participate in clinical trials? A mixed methods study | BMC Cancer | Full Text

Survival plots of time-to-event outcomes in clinical trials: good practice and pitfalls - The Lancet

Radiology workflow for RECIST assessment in clinical trials: Can we reconcile time-efficiency and quality? - European Journal of Radiology

EPIC: an evaluation of the psychological impact of early-phase clinical trials in cancer patients - ESMO Open
![PDF] Real-time clinical note monitoring to detect conditions for rapid follow-up: A case study of clinical trial enrollment in drug-induced torsades de pointes and Stevens-Johnson syndrome | Semantic Scholar PDF] Real-time clinical note monitoring to detect conditions for rapid follow-up: A case study of clinical trial enrollment in drug-induced torsades de pointes and Stevens-Johnson syndrome | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c0c7d1f9d77ad66be4c9dedda160509f83489880/3-Figure1-1.png)
PDF] Real-time clinical note monitoring to detect conditions for rapid follow-up: A case study of clinical trial enrollment in drug-induced torsades de pointes and Stevens-Johnson syndrome | Semantic Scholar

Schematic of data collection and timing. The initial therapy period for... | Download Scientific Diagram

An Application of the Patient Rule-Induction Method to Detect Clinically Meaningful Subgroups from Failed Phase III Clinical Trials

Protocol of the Definition for the Assessment of Time-to-event Endpoints in CANcer trials (DATECAN) project: Formal consensus method for the development of guidelines for standardised time-to-event endpoints' definitions in cancer clinical trials -