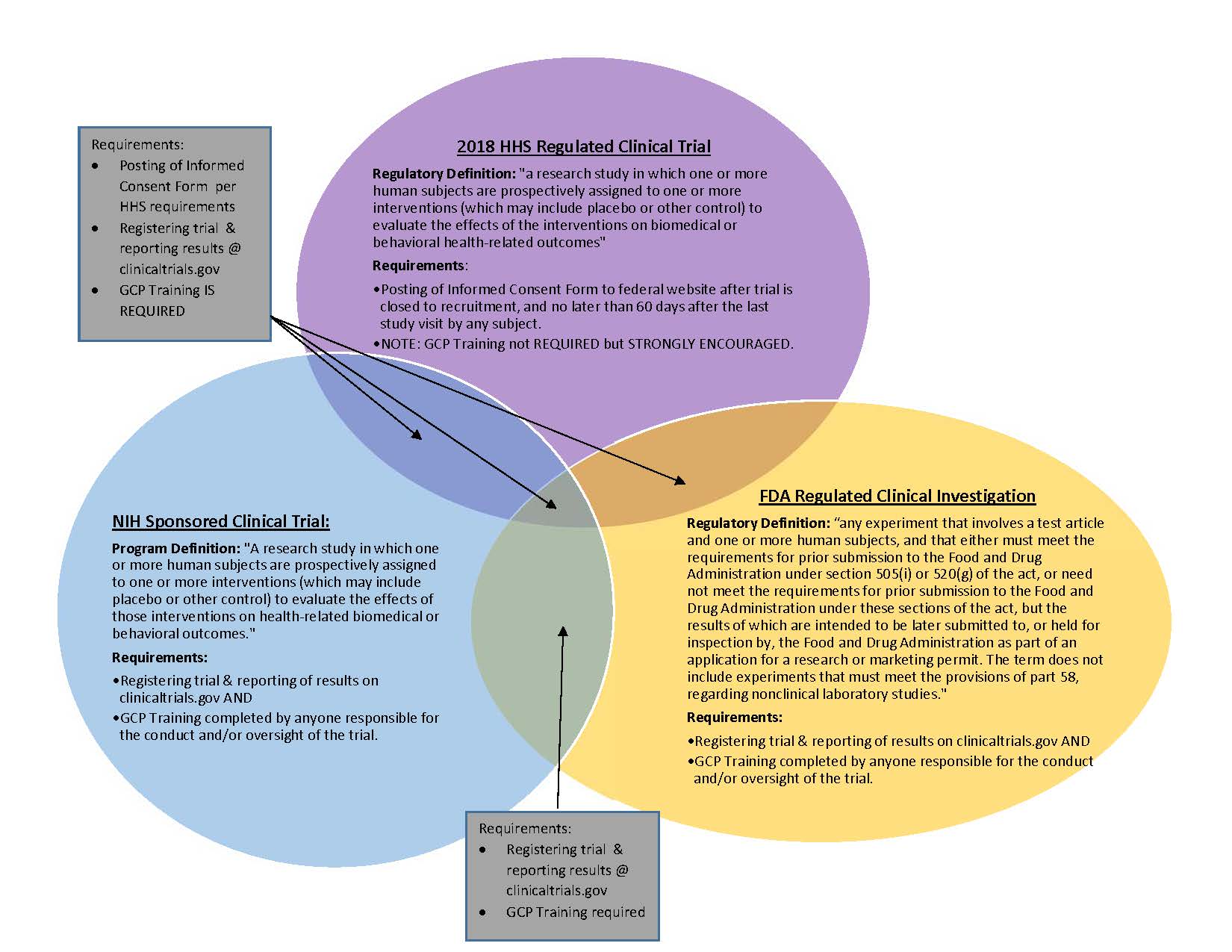
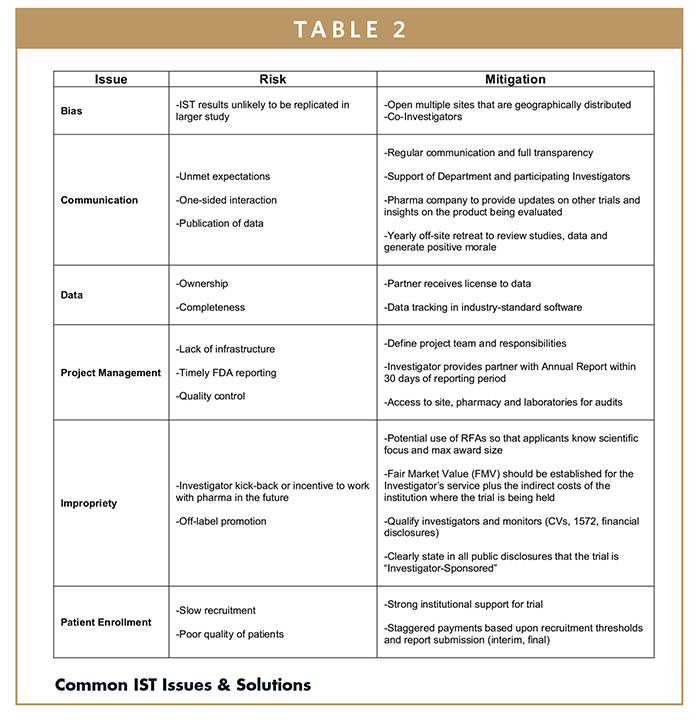
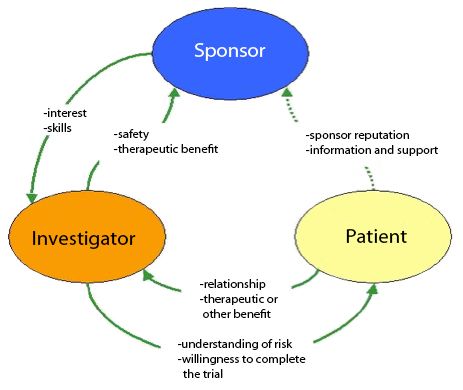
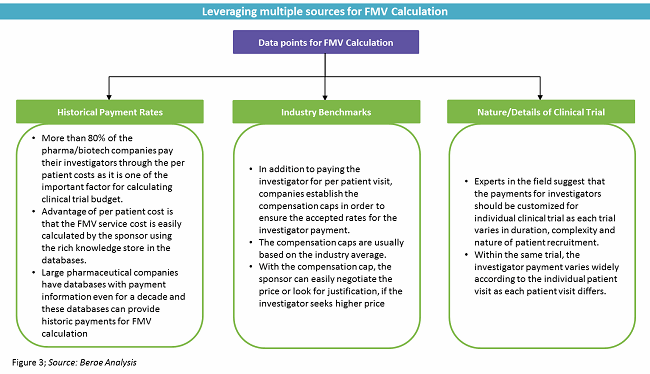
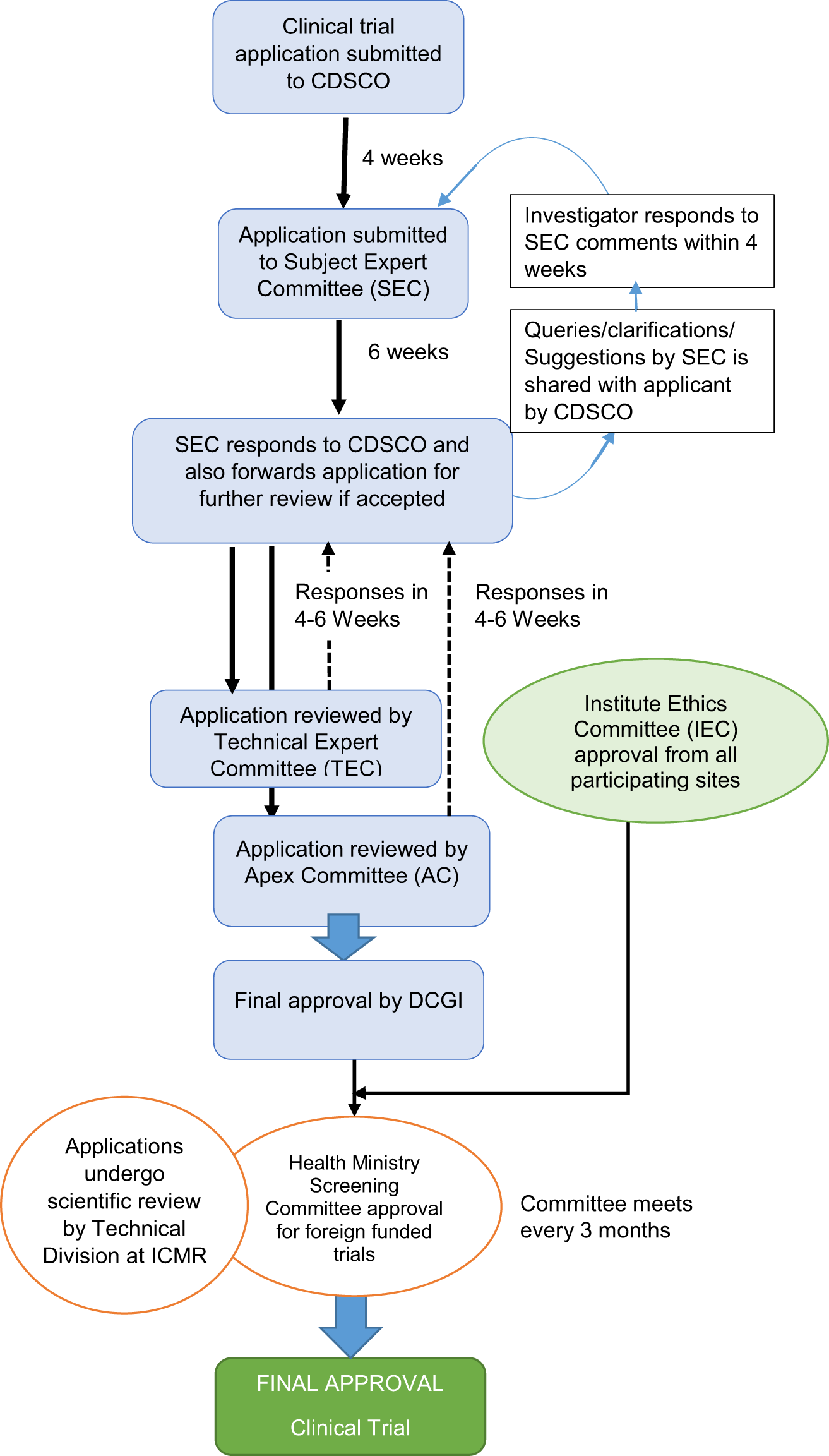
Frontiers | The Current Status and Future Direction of Clinical Research in Japan From a Regulatory Perspective
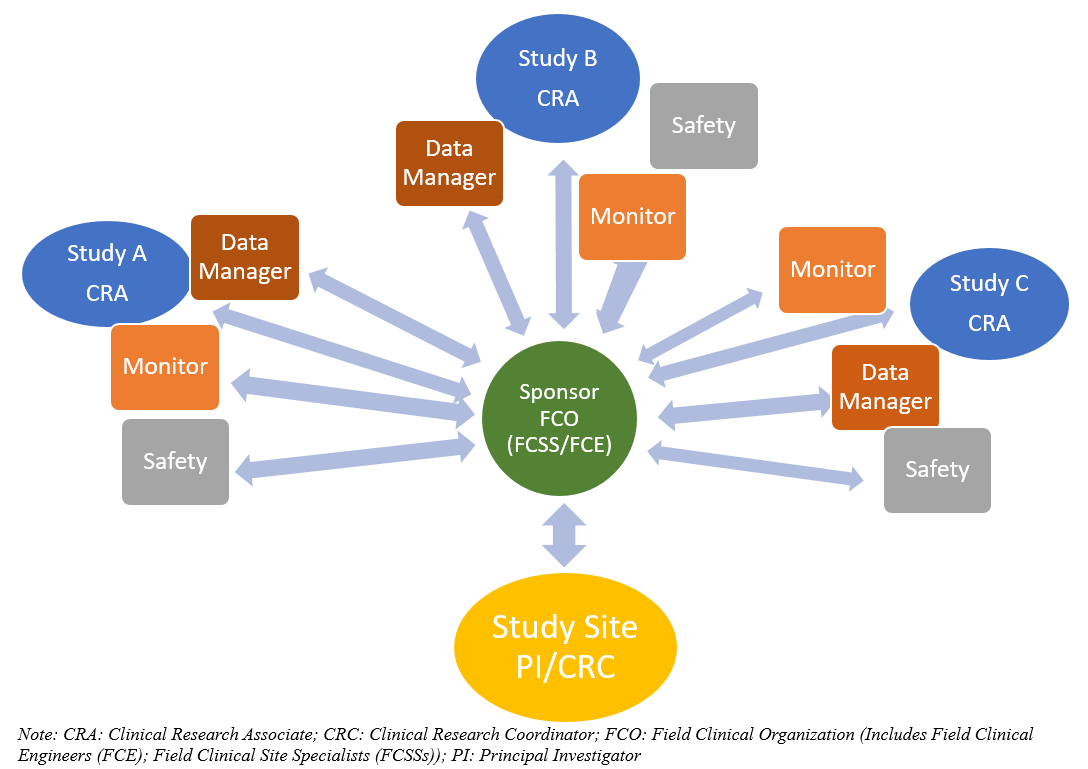
Sponsor-Site Communication in Device Trials: Evolution of a Dedicated Field Clinical Organization Throughout Study Execution - ACRP
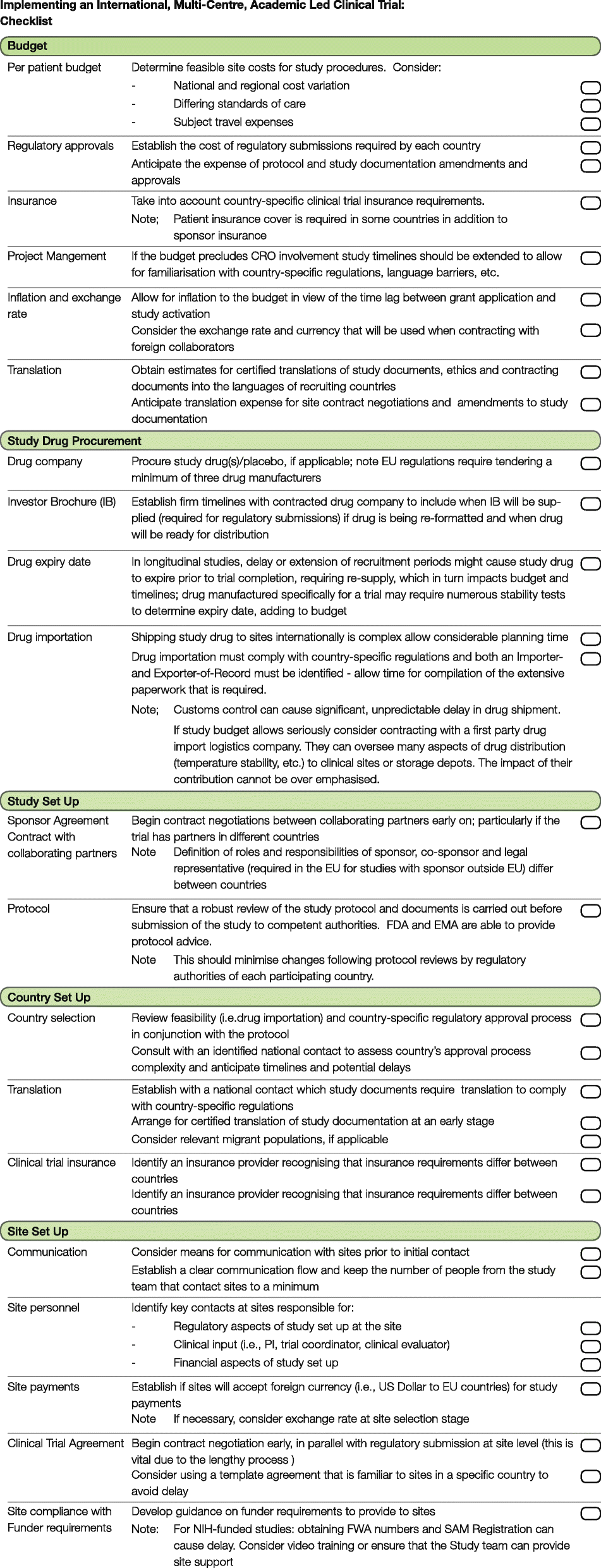
A checklist for clinical trials in rare disease: obstacles and anticipatory actions—lessons learned from the FOR-DMD trial | Trials | Full Text

Session III: (Breakout A) - Nuts and Bolts of Clinical Trials Participation for Investigators and Sub-Investigators Clinical Research Documentation. - ppt video online download
Issues, challenges, and the way forward in conducting clinical trials among neonates: investigators' perspective | Journal of Perinatology

WHEN, WHY AND HOW SPONSOR, CONTRACT RESEARCH ORGANISATION (CROs) AND RESEARCH SITES WORK TOGETHER: – your pharmacy guide

What is a CRO and how can it help you in your clinical trial - Leon Research | CRO - Clinical Trials Spain, Italy and Portugal
![PDF] Sponsor-investigator-relationship: challenges, recent regulatory developments and future legislative trends | Semantic Scholar PDF] Sponsor-investigator-relationship: challenges, recent regulatory developments and future legislative trends | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/cebe49113140c5242f8a80e21c7ff4cf13329a03/4-Table4-1.png)