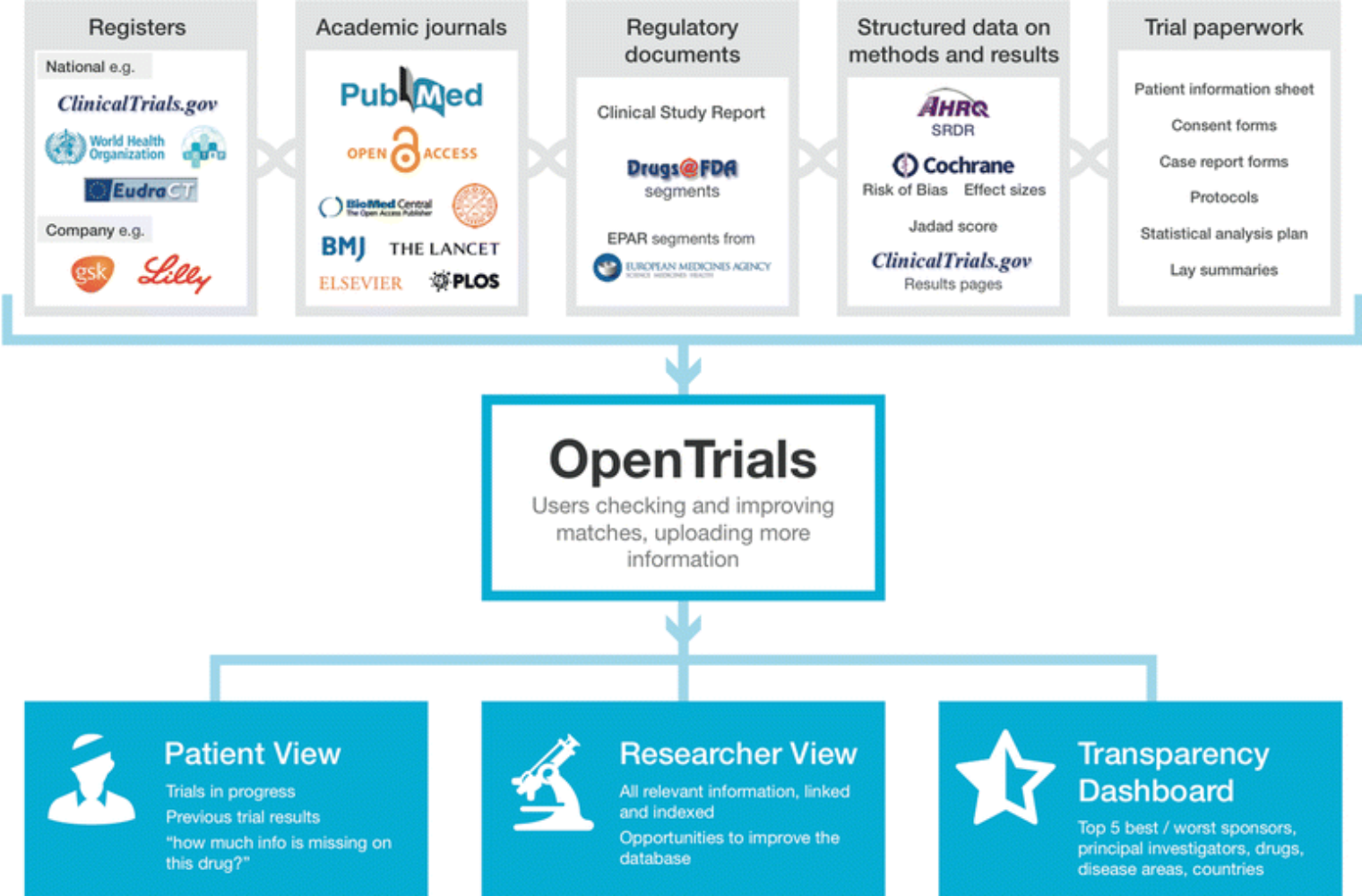
The WHO International Clinical Trials Registry Platform: Providing global clinical trial information to all - On Medicine

A schematic representation of the database for Aggregate Analysis of... | Download Scientific Diagram
![PDF] A database system for integrated clinical trial management, control, statistical analysis and ICH-compliant reporting | Semantic Scholar PDF] A database system for integrated clinical trial management, control, statistical analysis and ICH-compliant reporting | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b7222080945658c3980e16ccdbbceead72486f32/2-Figure1-1.png)
PDF] A database system for integrated clinical trial management, control, statistical analysis and ICH-compliant reporting | Semantic Scholar
The Database for Aggregate Analysis of ClinicalTrials.gov (AACT) and Subsequent Regrouping by Clinical Specialty | PLOS ONE
![PDF] An OMOP CDM-Based Relational Database of Clinical Research Eligibility Criteria | Semantic Scholar PDF] An OMOP CDM-Based Relational Database of Clinical Research Eligibility Criteria | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/78cbb6c6ecddffa5d21a86c3eaf862849b636105/10-Figure1-1.png)
PDF] An OMOP CDM-Based Relational Database of Clinical Research Eligibility Criteria | Semantic Scholar
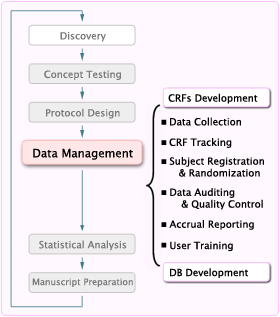
Understanding the process of Data Entry and Data Processing in Clinical Trials - Element Technologies

Statistical controversies in clinical research: limitations of open-label studies assessing antiangiogenic therapies with regard to evaluation of vascular adverse drug events—a meta-analysis - Annals of Oncology
![PDF] Utilization of a Clinical Trial Management System for the Whole Clinical Trial Process as an Integrated Database: System Development | Semantic Scholar PDF] Utilization of a Clinical Trial Management System for the Whole Clinical Trial Process as an Integrated Database: System Development | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b6a9ea604d7a322a539e1b7894536e2da07ce6a6/7-Figure2-1.png)