Clinical evaluation of semi-automatic open-source algorithmic software segmentation of the mandibular bone: Practical feasibility and assessment of a new course of action | PLOS ONE

Clinical Evaluation report of Existing data for CE-mark: review for regulatory professionals - YouTube

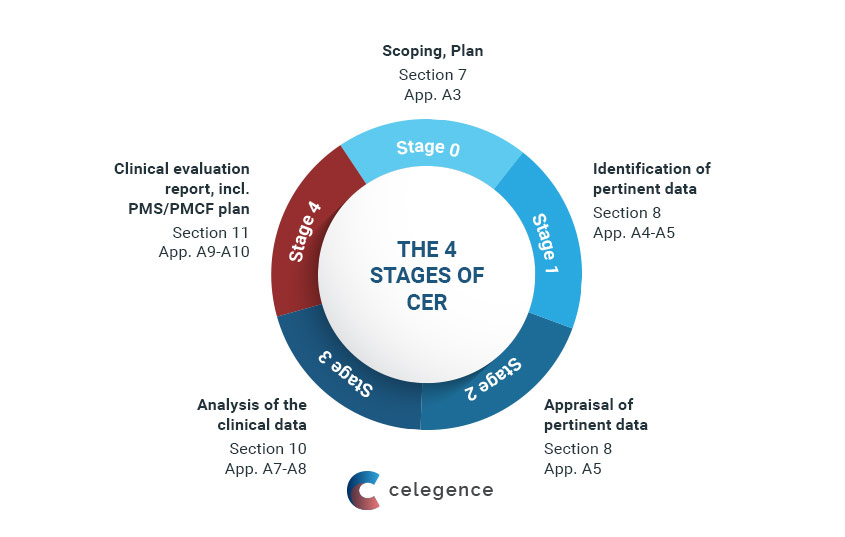
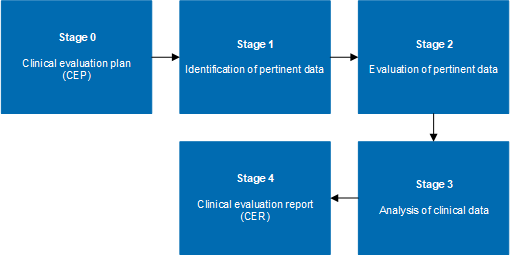
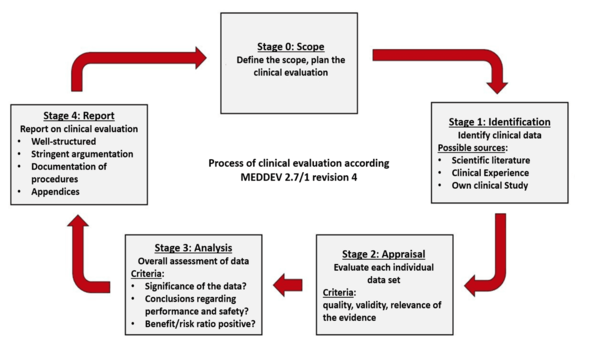
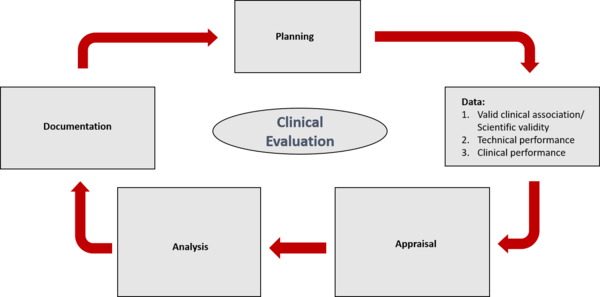
Clinical evaluation: is your Clinical Evaluation Report (CER) compliant with MEDDEV 2.7/1 rev. 4? - Thema Med