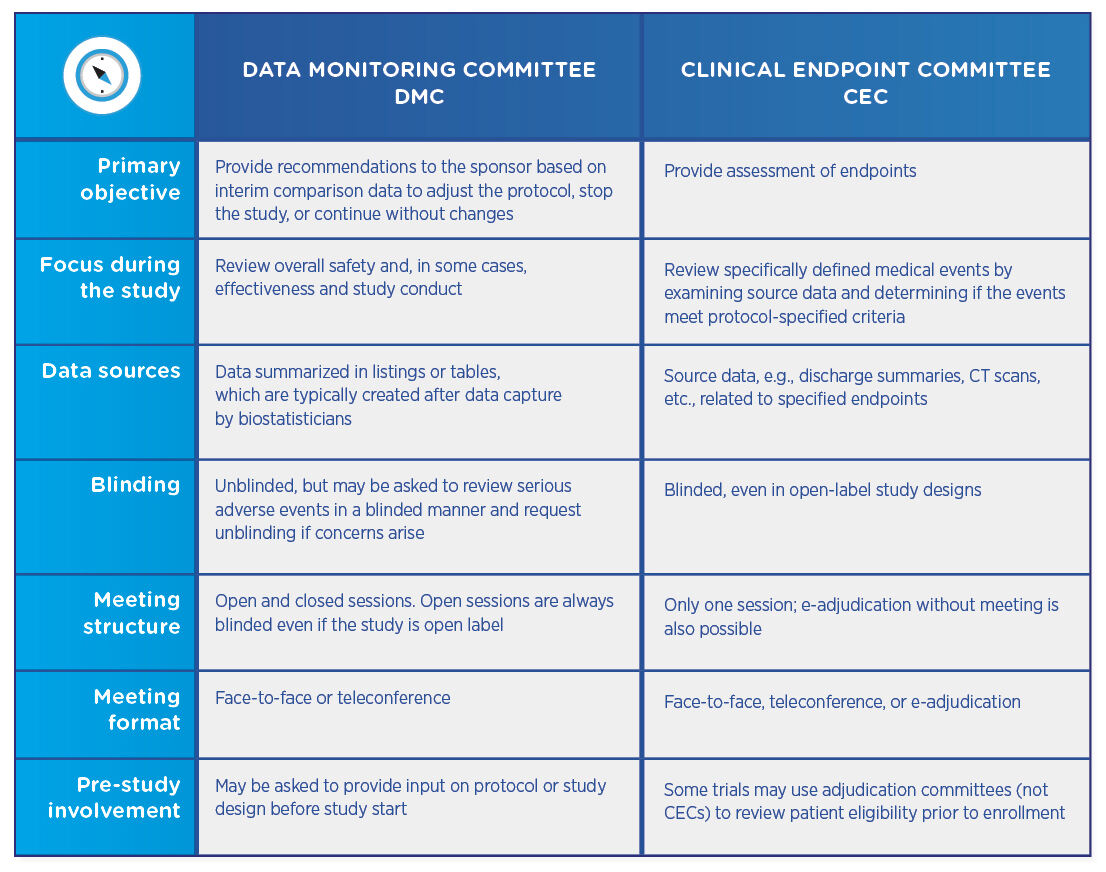
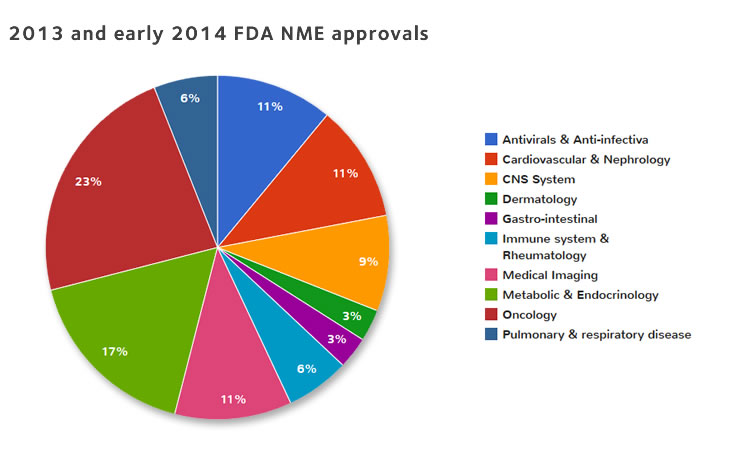
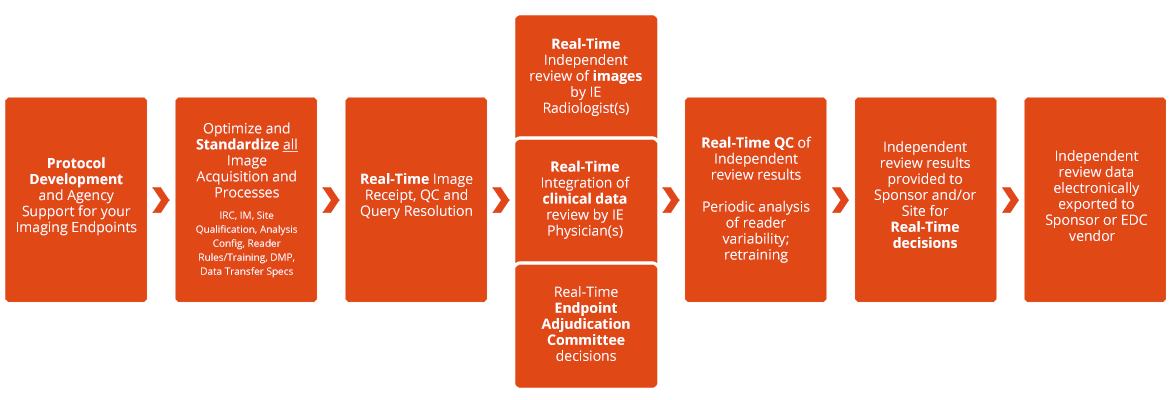
Clinical endpoint adjudication in a contemporary all-comers coronary stent investigation: Methodology and external validation - ScienceDirect
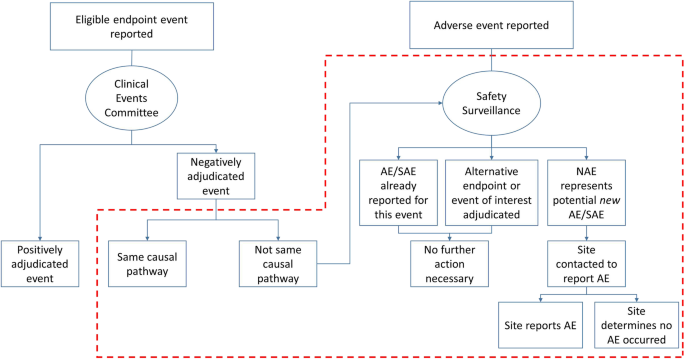
Methods for safety and endpoint ascertainment: identification of adverse events through scrutiny of negatively adjudicated events | Trials | Full Text

Guidelines for fracture healing assessments in clinical trials. Part I: Definitions and endpoint committees - Injury

Rationale and design of a prospective substudy of clinical endpoint adjudication processes within an investigator-reported randomised controlled trial in patients with coronary artery disease: the GLOBAL LEADERS Adjudication Sub-StudY (GLASSY) | BMJ
Inadequate planning and reporting of adjudication committees in clinical trials: Recommendation proposal