Teva Announces FDA Approval of AUSTEDO™ (deutetrabenazine) Tablets for the Treatment of Chorea Associated with Huntington's Disease — Hereditary Neurological Disease Centre

Teva launches first DTC for Austedo into competitive tardive dyskinesia 2-drug market | Fierce Pharma

HSG: Dr. Frank Discusses TEVA's New Drug AUSTEDO - Treatment of Chorea in Huntington's Disease - YouTube

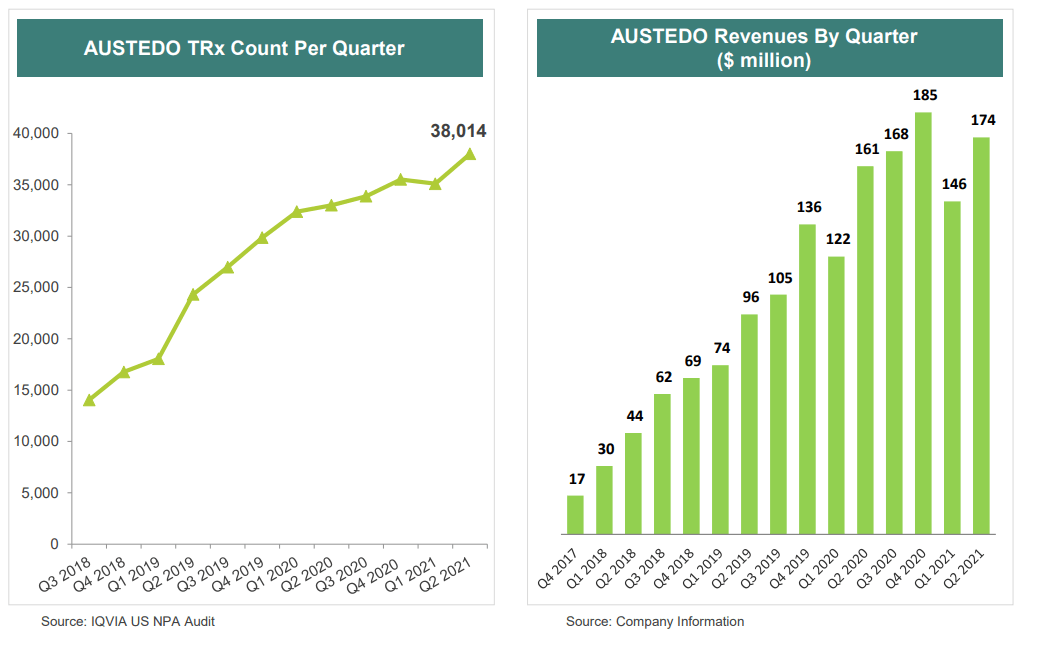
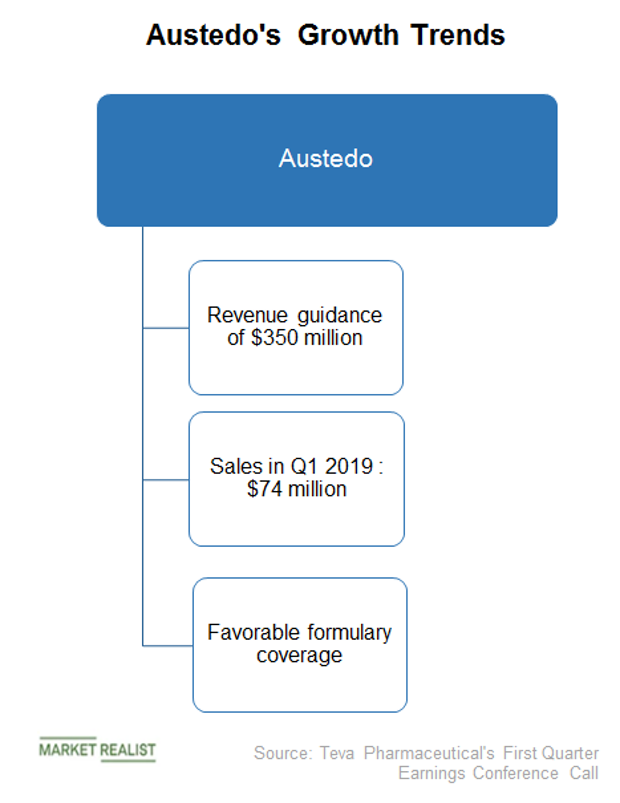
Punit on Twitter: "#Austedo stellar Q2 sales reported by Teva on 5th Aug'20 Austedo net sales in FY19 stood $412M & continues to grow in H1FY20👇 Q2-17- $1 M Q2-18- $44 M

Yo-Bio on Twitter: "$TEVA #Austedo have no black box warning for tardive dyskinesia. Only for HD (Read about HD and you will understand way). $NBIX #INGREZZA https://t.co/lXnoHCEUAA" / Twitter
Austedo (deutetrabenazine) for the Treatment of Chorea Associated with Huntington's Disease - Clinical Trials Arena